Haber-Bosch Process and Production of Fertilizers - UPSC Agriculture Allied Sector
What is Haber-Bosch Process and Production of Fertilizers in UPSC Agriculture Allied Sector?
Haber-Bosch Process and Production of Fertilizers is a key topic under Agriculture Allied Sector for UPSC Civil Services Examination. Key points include: The Haber-Bosch process synthesizes ammonia from atmospheric nitrogen and hydrogen.. It produces hundreds of millions of tonnes of reactive nitrogen for fertilizers annually.. Its output is comparable to or exceeds natural biological nitrogen fixation.. Understanding this topic is essential for both UPSC Prelims and Mains preparation.
Why is Haber-Bosch Process and Production of Fertilizers important for UPSC exam?
Haber-Bosch Process and Production of Fertilizers is a Medium-level topic in UPSC Agriculture Allied Sector. It is tested in both Prelims (factual MCQs) and Mains (analytical answer writing). Previous year UPSC questions have frequently covered aspects of Haber-Bosch Process and Production of Fertilizers, making it essential for comprehensive IAS preparation.
How to prepare Haber-Bosch Process and Production of Fertilizers for UPSC?
To prepare Haber-Bosch Process and Production of Fertilizers for UPSC: (1) Study the comprehensive notes covering all key concepts on Vaidra. (2) Practice previous year questions on this topic. (3) Connect it with current affairs using daily updates. (4) Revise using key takeaways and mind maps available for Agriculture Allied Sector. (5) Write practice answers linking Haber-Bosch Process and Production of Fertilizers to related GS Paper topics.
Key takeaways of Haber-Bosch Process and Production of Fertilizers for UPSC
- The Haber-Bosch process synthesizes ammonia from atmospheric nitrogen and hydrogen.
- It produces hundreds of millions of tonnes of reactive nitrogen for fertilizers annually.
- Its output is comparable to or exceeds natural biological nitrogen fixation.
- The process was critical for the Green Revolution, ensuring global food security.
- It is energy-intensive and contributes to greenhouse gas emissions and water pollution.
- Government policies like fertilizer subsidies and Neem-coated Urea aim to manage its use.

Haber-Bosch Process and Production of Fertilizers
📖 Introduction
💡 Key Takeaways
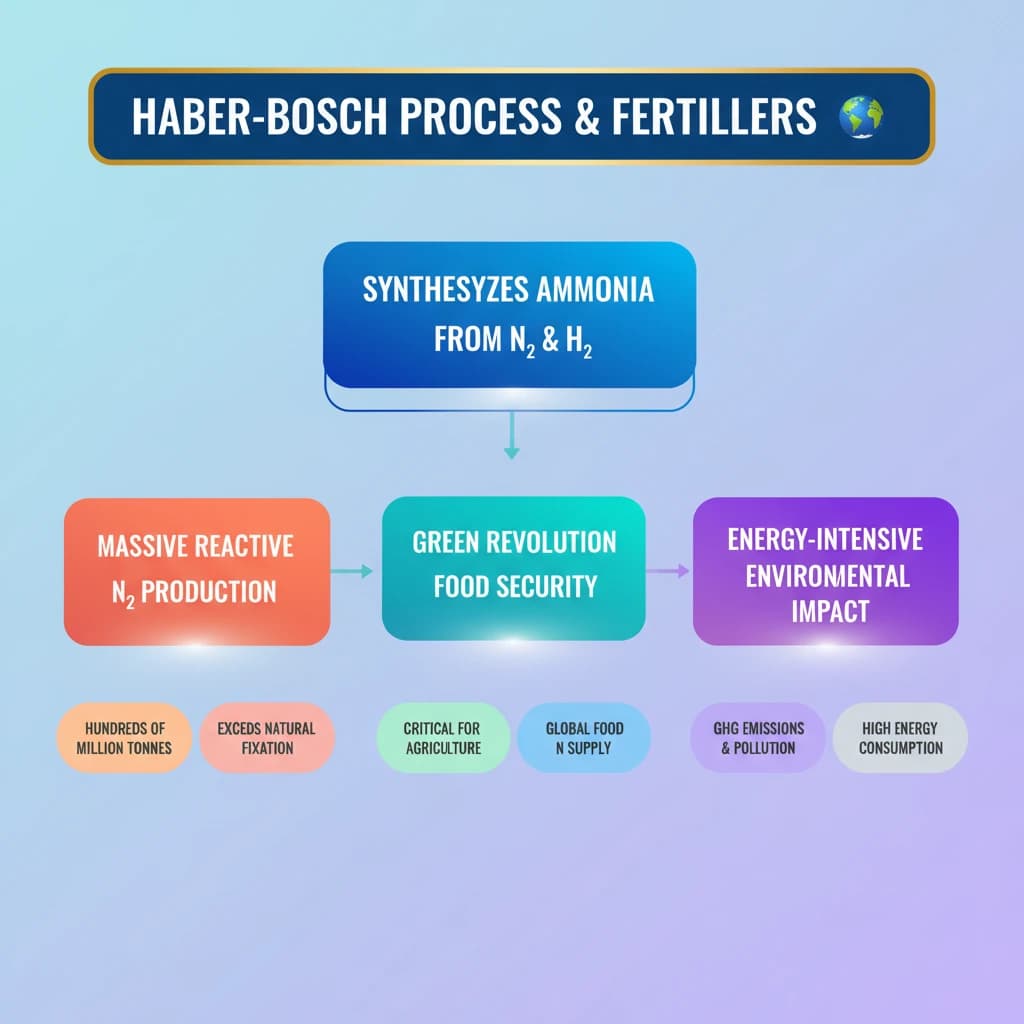
- •The Haber-Bosch process synthesizes ammonia from atmospheric nitrogen and hydrogen.
- •It produces hundreds of millions of tonnes of reactive nitrogen for fertilizers annually.
- •Its output is comparable to or exceeds natural biological nitrogen fixation.
- •The process was critical for the Green Revolution, ensuring global food security.
- •It is energy-intensive and contributes to greenhouse gas emissions and water pollution.
- •Government policies like fertilizer subsidies and Neem-coated Urea aim to manage its use.
🧠 Memory Techniques
