What is Ocean Acidification? - UPSC Environment And Ecology
What is What is Ocean Acidification? in UPSC Environment And Ecology?
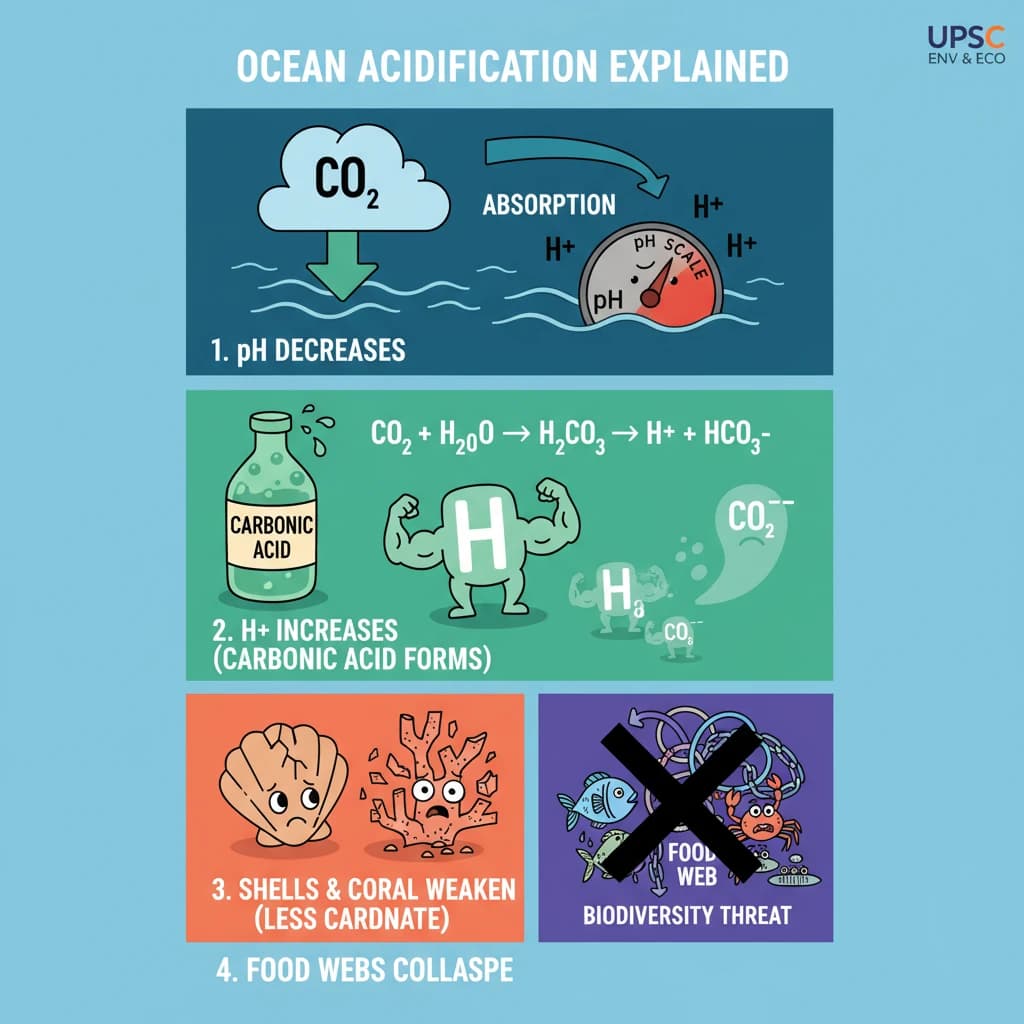
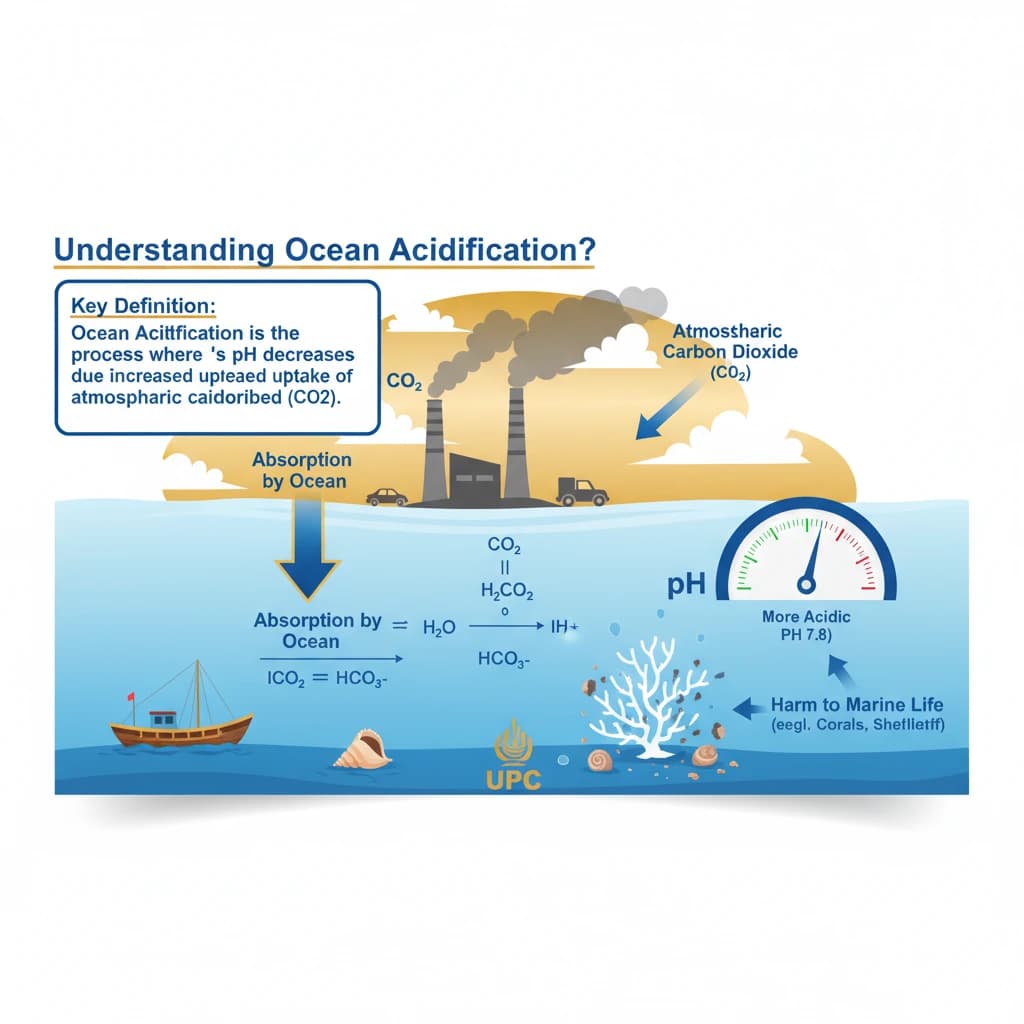
What is Ocean Acidification? is a key topic under Environment And Ecology for UPSC Civil Services Examination. Key points include: Ocean Acidification is the decrease in ocean pH due to excess atmospheric CO2 absorption.. CO2 reacts with seawater to form carbonic acid, releasing hydrogen ions and lowering pH.. This process reduces carbonate ions, essential for calcifying marine organisms like corals and shellfish.. Understanding this topic is essential for both UPSC Prelims and Mains preparation.
Why is What is Ocean Acidification? important for UPSC exam?
What is Ocean Acidification? is a Medium-level topic in UPSC Environment And Ecology. It is tested in both Prelims (factual MCQs) and Mains (analytical answer writing). Previous year UPSC questions have frequently covered aspects of What is Ocean Acidification?, making it essential for comprehensive IAS preparation.
How to prepare What is Ocean Acidification? for UPSC?
To prepare What is Ocean Acidification? for UPSC: (1) Study the comprehensive notes covering all key concepts on Vaidra. (2) Practice previous year questions on this topic. (3) Connect it with current affairs using daily updates. (4) Revise using key takeaways and mind maps available for Environment And Ecology. (5) Write practice answers linking What is Ocean Acidification? to related GS Paper topics.
Key takeaways of What is Ocean Acidification? for UPSC
- Ocean Acidification is the decrease in ocean pH due to excess atmospheric CO2 absorption.
- CO2 reacts with seawater to form carbonic acid, releasing hydrogen ions and lowering pH.
- This process reduces carbonate ions, essential for calcifying marine organisms like corals and shellfish.
- Impacts include weaker shells, reduced growth, and threats to marine food webs and biodiversity.
- Driven by anthropogenic CO2 emissions, it poses a significant threat to marine ecosystems, food security, and coastal economies.
- It is a critical aspect of climate change, demanding urgent global action to reduce emissions.
What is Ocean Acidification?
📖 Introduction

💡 Key Takeaways
- •Ocean Acidification is the decrease in ocean pH due to excess atmospheric CO2 absorption.
- •CO2 reacts with seawater to form carbonic acid, releasing hydrogen ions and lowering pH.
- •This process reduces carbonate ions, essential for calcifying marine organisms like corals and shellfish.
- •Impacts include weaker shells, reduced growth, and threats to marine food webs and biodiversity.
- •Driven by anthropogenic CO2 emissions, it poses a significant threat to marine ecosystems, food security, and coastal economies.
- •It is a critical aspect of climate change, demanding urgent global action to reduce emissions.
🧠 Memory Techniques