Causes of Acid Rain - UPSC Environment And Ecology
What is Causes of Acid Rain in UPSC Environment And Ecology?
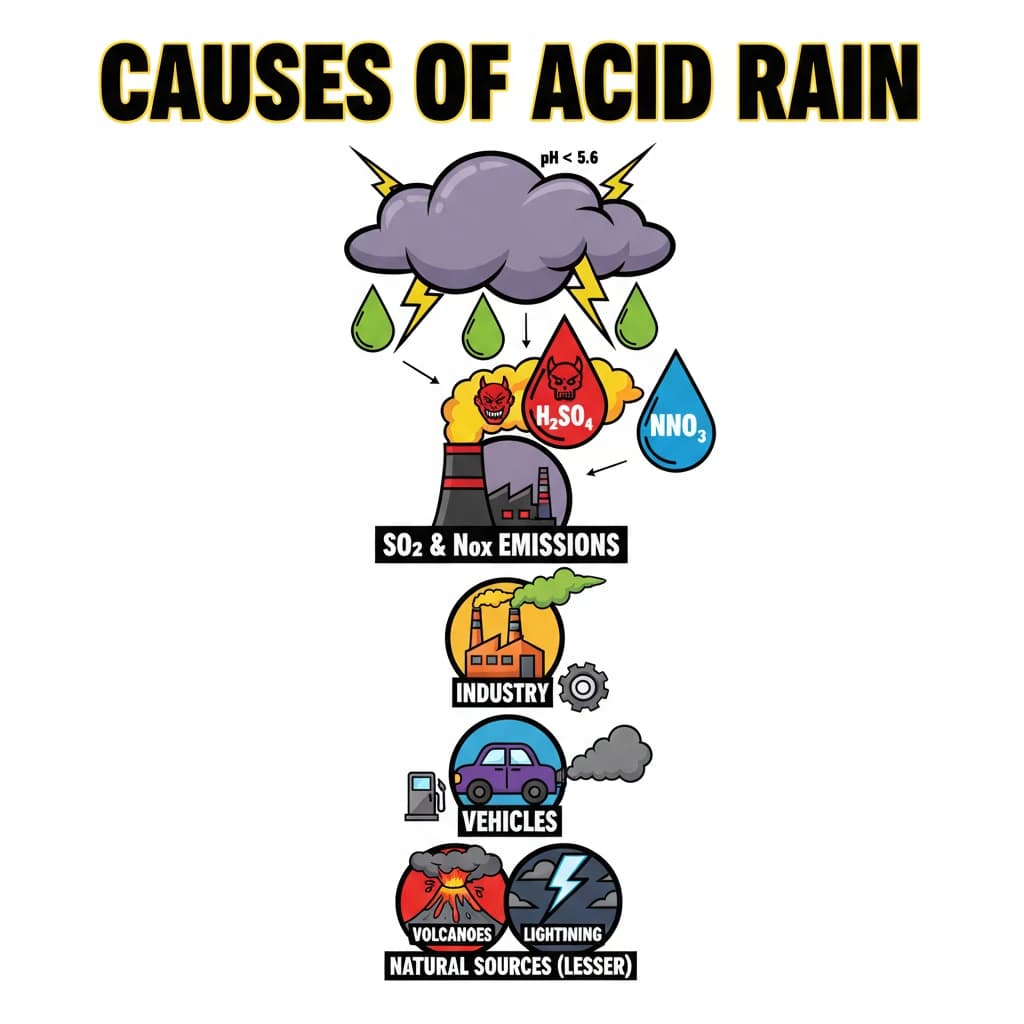
Causes of Acid Rain is a key topic under Environment And Ecology for UPSC Civil Services Examination. Key points include: Acid rain is precipitation with high levels of sulfuric and nitric acids, primarily caused by atmospheric pollutants.. The main anthropogenic causes are emissions of Sulfur Dioxide (SO2) and Nitrogen Oxides (NOx) from fossil fuel combustion.. Key sources include coal-fired power plants, industrial processes, and vehicle exhaust.. Understanding this topic is essential for both UPSC Prelims and Mains preparation.
Why is Causes of Acid Rain important for UPSC exam?
Causes of Acid Rain is a Medium-level topic in UPSC Environment And Ecology. It is tested in both Prelims (factual MCQs) and Mains (analytical answer writing). Previous year UPSC questions have frequently covered aspects of Causes of Acid Rain, making it essential for comprehensive IAS preparation.
How to prepare Causes of Acid Rain for UPSC?
To prepare Causes of Acid Rain for UPSC: (1) Study the comprehensive notes covering all key concepts on Vaidra. (2) Practice previous year questions on this topic. (3) Connect it with current affairs using daily updates. (4) Revise using key takeaways and mind maps available for Environment And Ecology. (5) Write practice answers linking Causes of Acid Rain to related GS Paper topics.
Key takeaways of Causes of Acid Rain for UPSC
- Acid rain is precipitation with high levels of sulfuric and nitric acids, primarily caused by atmospheric pollutants.
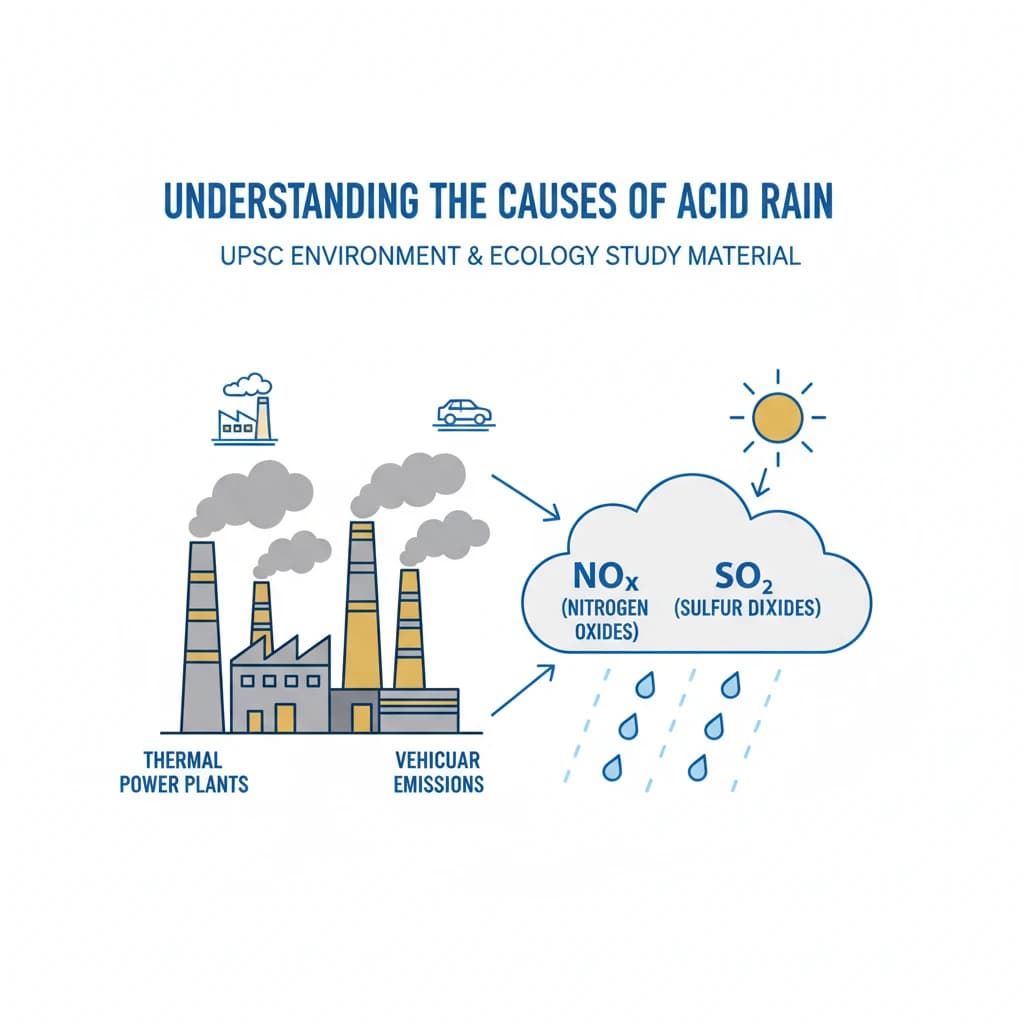
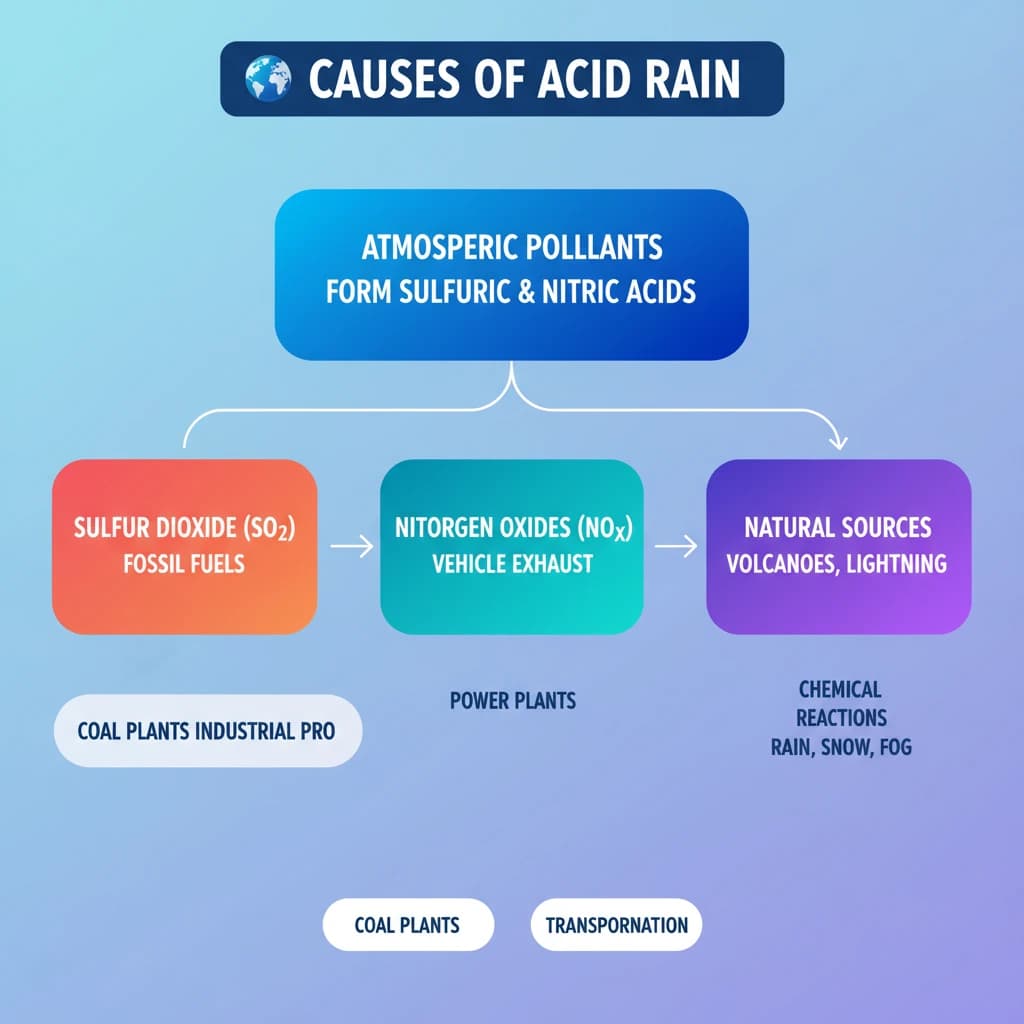
- The main anthropogenic causes are emissions of Sulfur Dioxide (SO2) and Nitrogen Oxides (NOx) from fossil fuel combustion.
- Key sources include coal-fired power plants, industrial processes, and vehicle exhaust.
- Natural sources like volcanic eruptions and lightning also contribute, but to a lesser extent.
- SO2 and NOx undergo chemical reactions in the atmosphere to form sulfuric and nitric acids, which then fall as acid rain.
- Acid rain has severe impacts on ecosystems, infrastructure, and human health, necessitating global and local mitigation efforts.
Causes of Acid Rain
📖 Introduction
💡 Key Takeaways
- •Acid rain is precipitation with high levels of sulfuric and nitric acids, primarily caused by atmospheric pollutants.
- •The main anthropogenic causes are emissions of Sulfur Dioxide (SO2) and Nitrogen Oxides (NOx) from fossil fuel combustion.
- •Key sources include coal-fired power plants, industrial processes, and vehicle exhaust.
- •Natural sources like volcanic eruptions and lightning also contribute, but to a lesser extent.
- •SO2 and NOx undergo chemical reactions in the atmosphere to form sulfuric and nitric acids, which then fall as acid rain.
- •Acid rain has severe impacts on ecosystems, infrastructure, and human health, necessitating global and local mitigation efforts.
🧠 Memory Techniques