What are the Key Facts about Lithium? - UPSC Environment And Ecology
What is What are the Key Facts about Lithium? in UPSC Environment And Ecology?
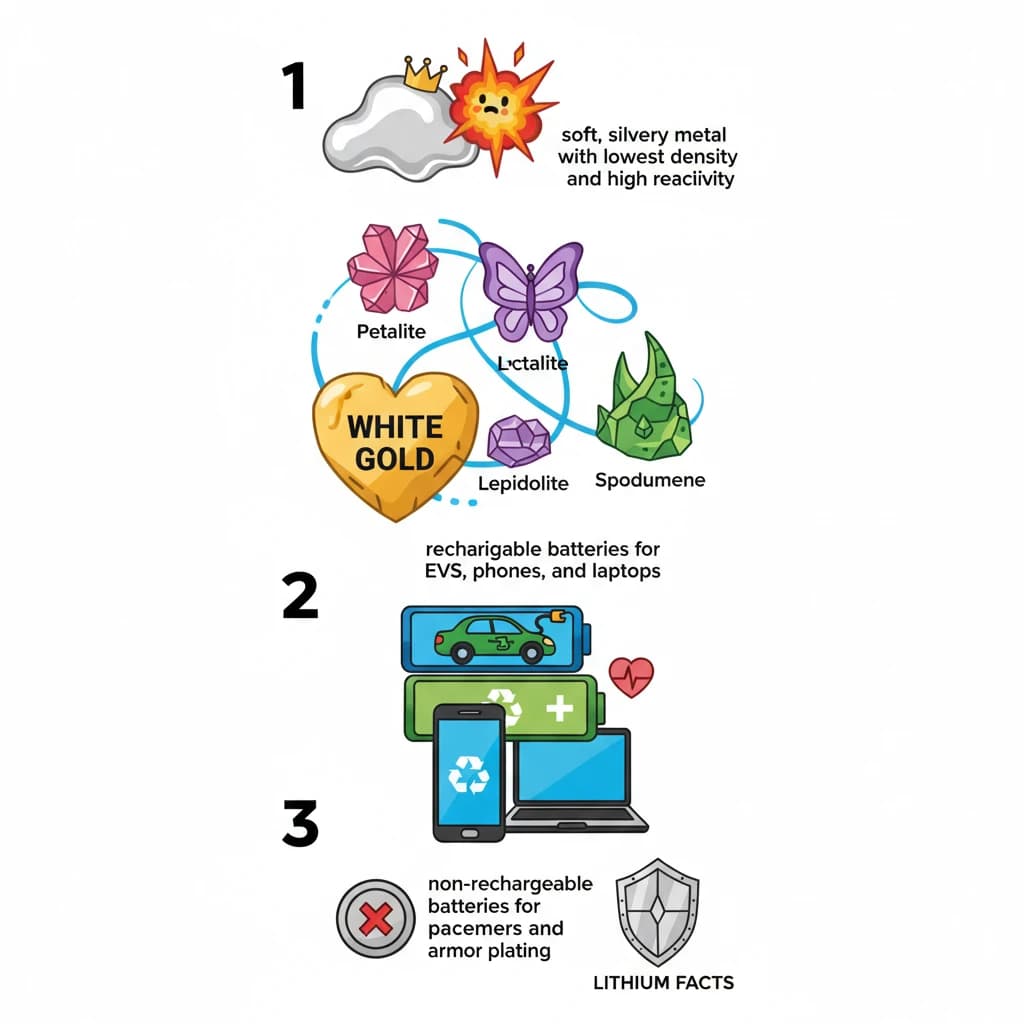
What are the Key Facts about Lithium? is a key topic under Environment And Ecology for UPSC Civil Services Examination. Key points include: Lithium is a soft, silvery metal with the lowest density and high reactivity.. Key ores include Petalite, Lepidolite, and Spodumene, often called 'white gold'.. Primarily used in rechargeable batteries for EVs, phones, and laptops.. Understanding this topic is essential for both UPSC Prelims and Mains preparation.
Why is What are the Key Facts about Lithium? important for UPSC exam?
What are the Key Facts about Lithium? is a Medium-level topic in UPSC Environment And Ecology. It is tested in both Prelims (factual MCQs) and Mains (analytical answer writing). Previous year UPSC questions have frequently covered aspects of What are the Key Facts about Lithium?, making it essential for comprehensive IAS preparation.
How to prepare What are the Key Facts about Lithium? for UPSC?
To prepare What are the Key Facts about Lithium? for UPSC: (1) Study the comprehensive notes covering all key concepts on Vaidra. (2) Practice previous year questions on this topic. (3) Connect it with current affairs using daily updates. (4) Revise using key takeaways and mind maps available for Environment And Ecology. (5) Write practice answers linking What are the Key Facts about Lithium? to related GS Paper topics.
Key takeaways of What are the Key Facts about Lithium? for UPSC
- Lithium is a soft, silvery metal with the lowest density and high reactivity.
- Key ores include Petalite, Lepidolite, and Spodumene, often called 'white gold'.
- Primarily used in rechargeable batteries for EVs, phones, and laptops.
- Also used in non-rechargeable batteries for pacemakers and in lightweight alloys for armor plating.
- A critical mineral vital for global energy transition, EVs, and renewable energy storage.
- India's domestic Lithium discoveries are strategically important for self-reliance.

What are the Key Facts about Lithium?
📖 Introduction

💡 Key Takeaways
- •Lithium is a soft, silvery metal with the lowest density and high reactivity.
- •Key ores include Petalite, Lepidolite, and Spodumene, often called 'white gold'.
- •Primarily used in rechargeable batteries for EVs, phones, and laptops.
- •Also used in non-rechargeable batteries for pacemakers and in lightweight alloys for armor plating.
- •A critical mineral vital for global energy transition, EVs, and renewable energy storage.
- •India's domestic Lithium discoveries are strategically important for self-reliance.
🧠 Memory Techniques