What is Nitrogen Pollution? - UPSC Environment And Ecology
What is What is Nitrogen Pollution? in UPSC Environment And Ecology?
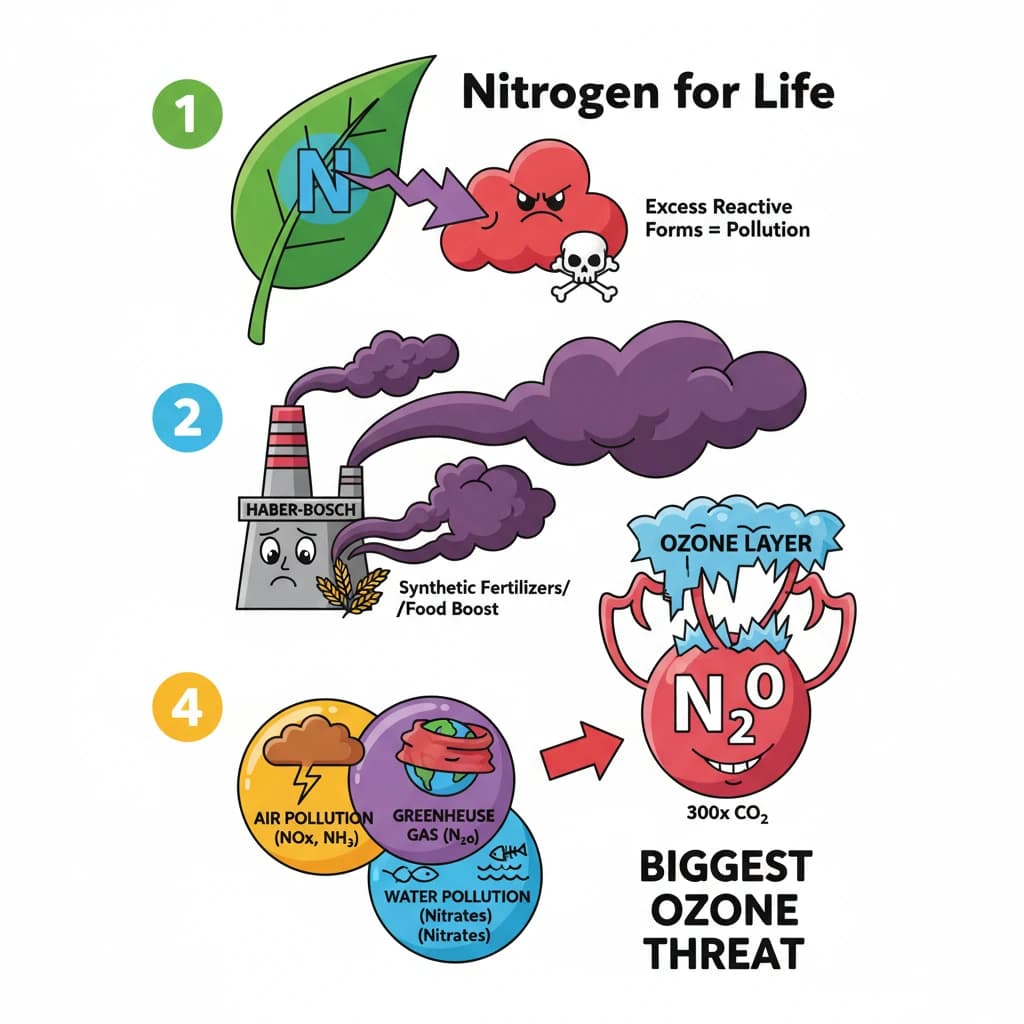
What is Nitrogen Pollution? is a key topic under Environment And Ecology for UPSC Civil Services Examination. Key points include: Nitrogen is essential for life but its reactive forms, when in excess, cause significant pollution.. The Haber-Bosch process enabled synthetic fertilizers, boosting food but also reactive nitrogen flows.. Nitrogen pollution manifests as air pollution (NOx, NH₃), greenhouse gas (N₂O), and water pollution (nitrates).. Understanding this topic is essential for both UPSC Prelims and Mains preparation.
Why is What is Nitrogen Pollution? important for UPSC exam?
What is Nitrogen Pollution? is a Medium-level topic in UPSC Environment And Ecology. It is tested in both Prelims (factual MCQs) and Mains (analytical answer writing). Previous year UPSC questions have frequently covered aspects of What is Nitrogen Pollution?, making it essential for comprehensive IAS preparation.
How to prepare What is Nitrogen Pollution? for UPSC?
To prepare What is Nitrogen Pollution? for UPSC: (1) Study the comprehensive notes covering all key concepts on Vaidra. (2) Practice previous year questions on this topic. (3) Connect it with current affairs using daily updates. (4) Revise using key takeaways and mind maps available for Environment And Ecology. (5) Write practice answers linking What is Nitrogen Pollution? to related GS Paper topics.
Key takeaways of What is Nitrogen Pollution? for UPSC
- Nitrogen is essential for life but its reactive forms, when in excess, cause significant pollution.
- The Haber-Bosch process enabled synthetic fertilizers, boosting food but also reactive nitrogen flows.
- Nitrogen pollution manifests as air pollution (NOx, NH₃), greenhouse gas (N₂O), and water pollution (nitrates).
- Nitrous oxide (N₂O) is a powerful GHG (300x CO₂) and the largest human threat to the ozone layer.
- Effects include global warming, ozone depletion, biodiversity loss (dead zones, acidic soils), and severe air quality degradation (smog, particulates).

What is Nitrogen Pollution?
📖 Introduction
💡 Key Takeaways
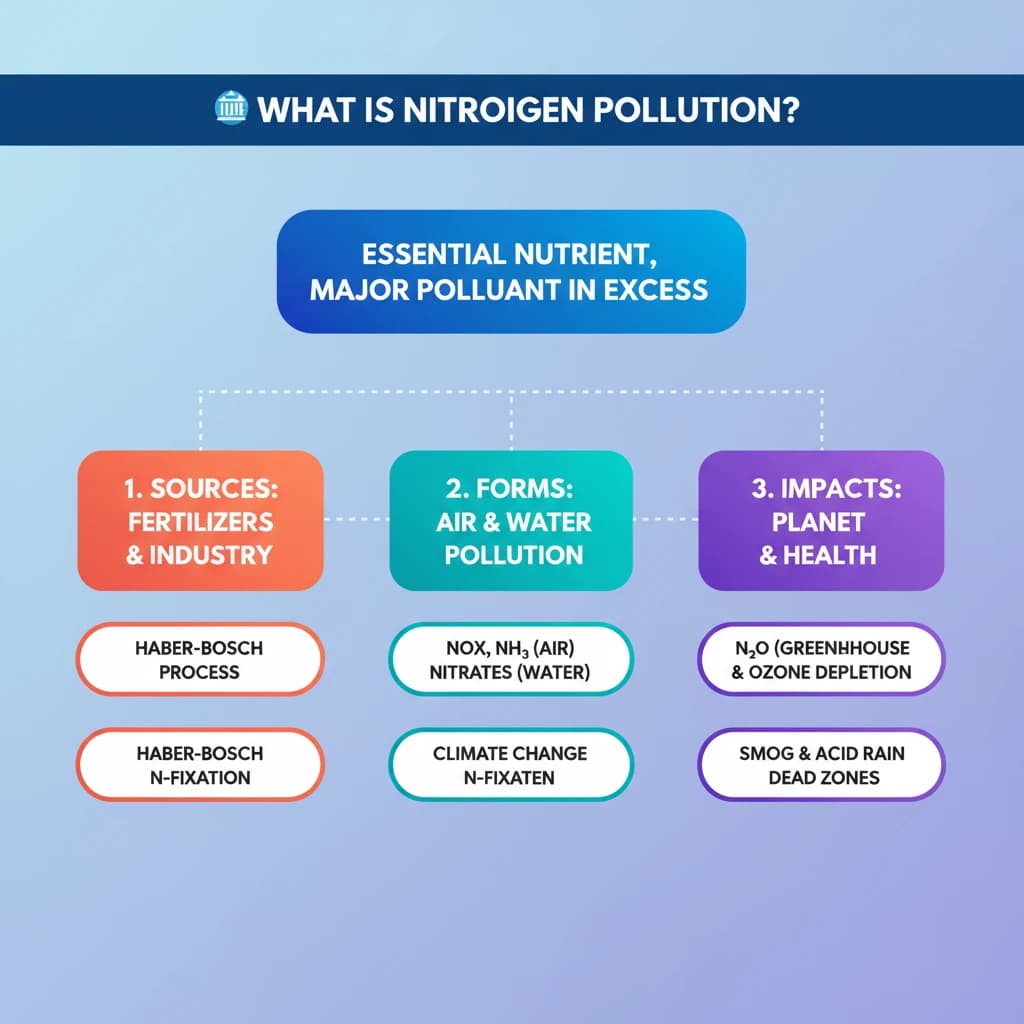
- •Nitrogen is essential for life but its reactive forms, when in excess, cause significant pollution.
- •The Haber-Bosch process enabled synthetic fertilizers, boosting food but also reactive nitrogen flows.
- •Nitrogen pollution manifests as air pollution (NOx, NH₃), greenhouse gas (N₂O), and water pollution (nitrates).
- •Nitrous oxide (N₂O) is a powerful GHG (300x CO₂) and the largest human threat to the ozone layer.
- •Effects include global warming, ozone depletion, biodiversity loss (dead zones, acidic soils), and severe air quality degradation (smog, particulates).
🧠 Memory Techniques