CAR-T Cell Therapy - UPSC Science And Technology
What is CAR-T Cell Therapy in UPSC Science And Technology?
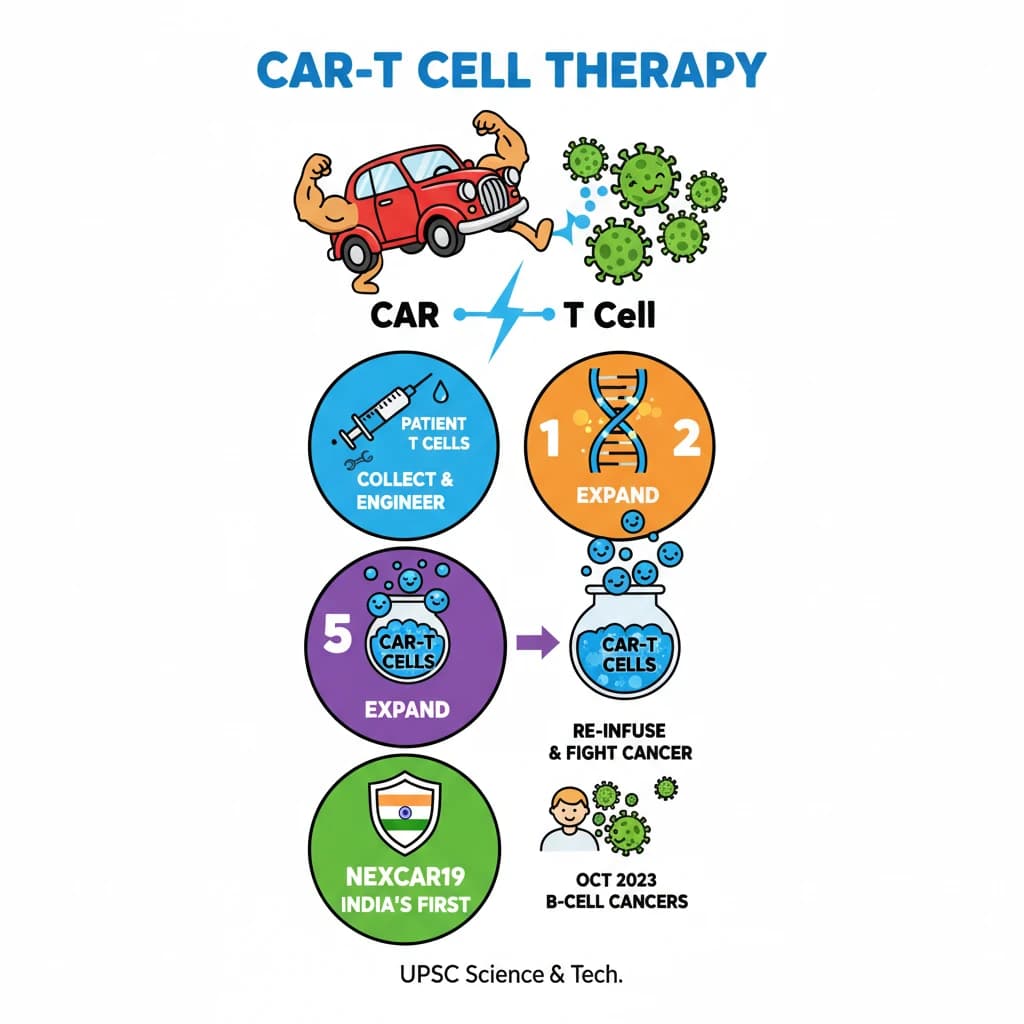
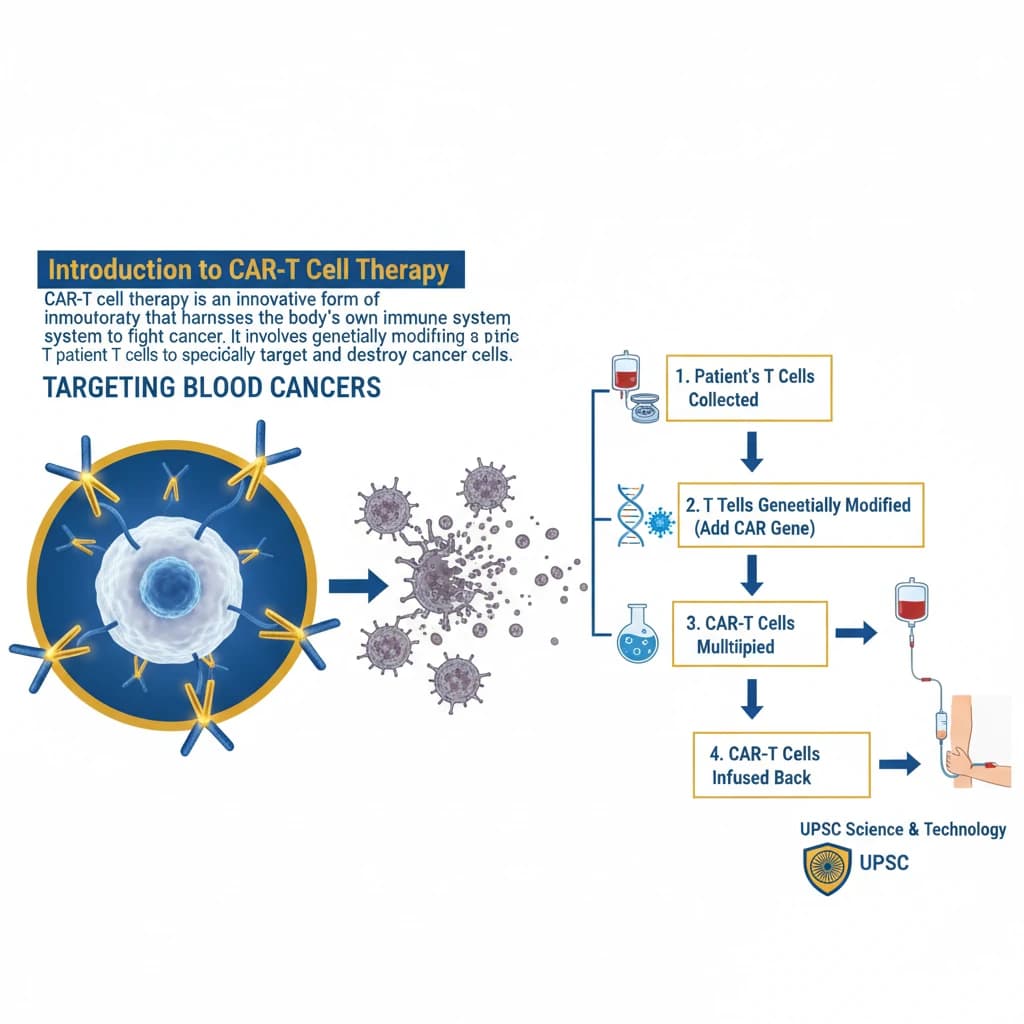
CAR-T Cell Therapy is a key topic under Science And Technology for UPSC Civil Services Examination. Key points include: CAR-T cell therapy uses genetically modified T cells to fight cancer.. It involves collecting patient T cells, engineering them with a Chimeric Antigen Receptor (CAR), expanding them, and re-infusing them.. CARs are designed to recognize specific antigens on cancer cells.. Understanding this topic is essential for both UPSC Prelims and Mains preparation.
Why is CAR-T Cell Therapy important for UPSC exam?
CAR-T Cell Therapy is a Medium-level topic in UPSC Science And Technology. It is tested in both Prelims (factual MCQs) and Mains (analytical answer writing). Previous year UPSC questions have frequently covered aspects of CAR-T Cell Therapy, making it essential for comprehensive IAS preparation.
How to prepare CAR-T Cell Therapy for UPSC?
To prepare CAR-T Cell Therapy for UPSC: (1) Study the comprehensive notes covering all key concepts on Vaidra. (2) Practice previous year questions on this topic. (3) Connect it with current affairs using daily updates. (4) Revise using key takeaways and mind maps available for Science And Technology. (5) Write practice answers linking CAR-T Cell Therapy to related GS Paper topics.
Key takeaways of CAR-T Cell Therapy for UPSC
- CAR-T cell therapy uses genetically modified T cells to fight cancer.
- It involves collecting patient T cells, engineering them with a Chimeric Antigen Receptor (CAR), expanding them, and re-infusing them.
- CARs are designed to recognize specific antigens on cancer cells.
- NexCAR19 is India's first indigenous CAR-T therapy, approved in October 2023 for B-cell cancers.
- This therapy represents a major advancement in precision medicine and India's self-reliance in advanced healthcare.
CAR-T Cell Therapy
📖 Introduction
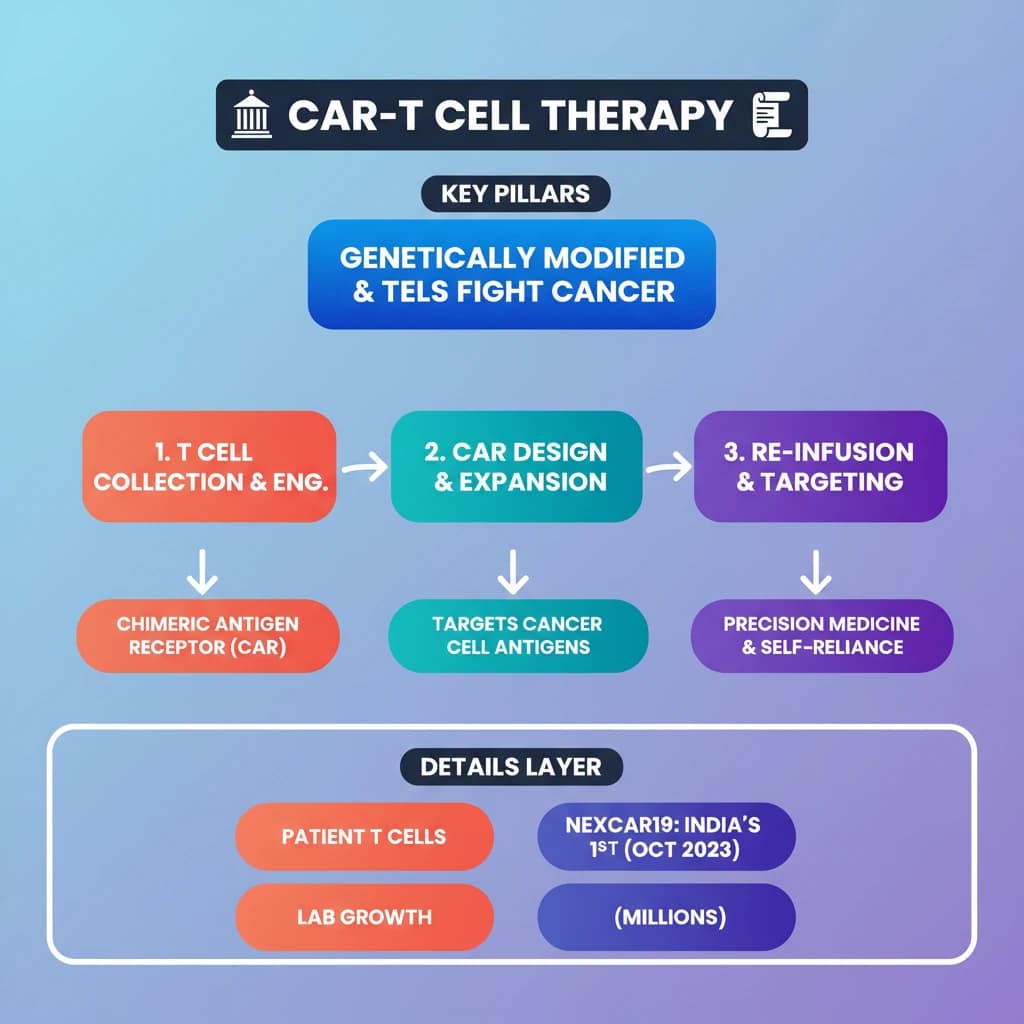
💡 Key Takeaways
- •CAR-T cell therapy uses genetically modified T cells to fight cancer.
- •It involves collecting patient T cells, engineering them with a Chimeric Antigen Receptor (CAR), expanding them, and re-infusing them.
- •CARs are designed to recognize specific antigens on cancer cells.
- •NexCAR19 is India's first indigenous CAR-T therapy, approved in October 2023 for B-cell cancers.
- •This therapy represents a major advancement in precision medicine and India's self-reliance in advanced healthcare.
🧠 Memory Techniques